REVOLUTIONARY SOLUTIONS
Unique product on the basis of bacteriophages
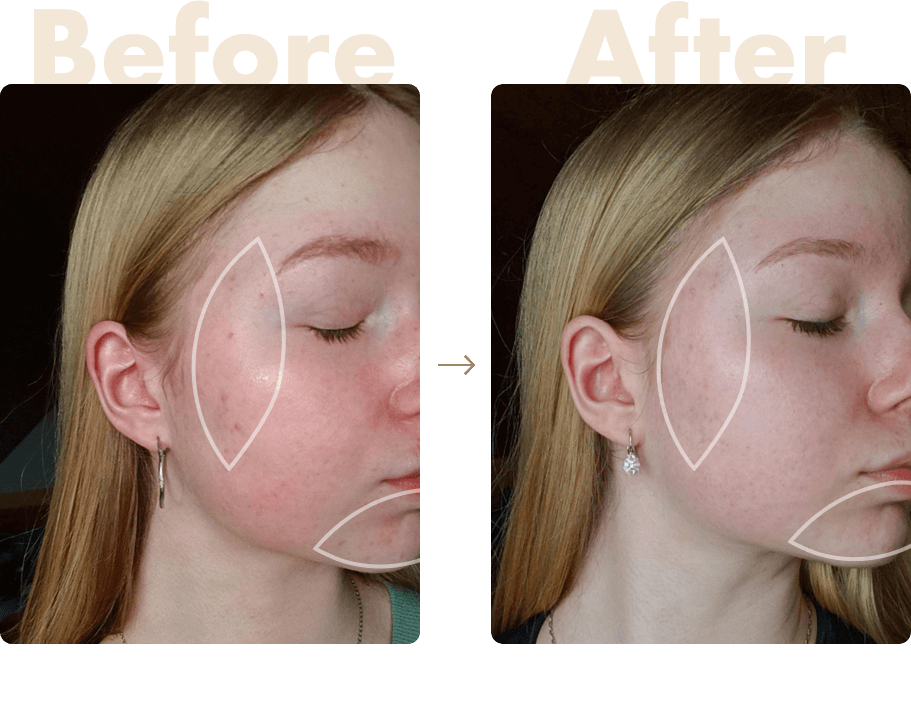
Isolated phages kill the bacteria associated with acne and problematic skin. This product is considered revolutionary due to the high stability and desired genomic features of the bacteriophages. Phage therapy has been proved to be one of the most effective methods how to fight against the resistant strains compared to antibiotics.
 Aloe vera
Aloe vera Lavandula Angustifolia
Lavandula Angustifolia
![]()
Bioactive gel
Revolution in the fight against problematic skin on the basis of bacteriophage which don’t irritate your skin but help its hydration and beautiful natural look.
Buy 22,96 €
![]()
Skin Spray
A special cosmetic product designed for eczematous skin. Bacteriophages are able to multiply at the site of infection without burdening the patient's condition or its microflora.
Buy 24,92 €